Preferred Buyer From :
All over the world
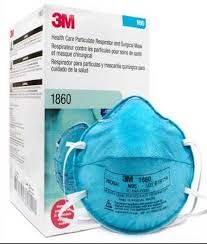
We offer N95 Particulate Respirators.
Specifications
Exceeds test requirements of Title 42, Code of Federal Regulations, Part 84 (42 CFR 84)
Meets performance criteria recommended by CDC for respiratory devices used in healthcare settings for protection against Mycobacterium tuberculosis (MTB), the infectious agent that causes tuberculosis (TB).
Particulates for which these respirators provide protection include contaminants such as dusts, fumes, mists and smoke.
N Series respirators are intended for use in those workplaces free of oil aerosols