Listing ID #5110013
Company Information
Ask for more detail from the seller
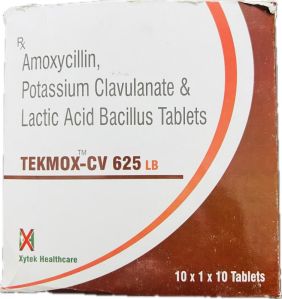
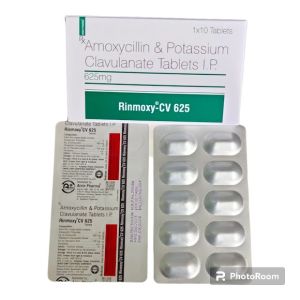
Contact Suppliereach film-coated tablet contains amoxicillin trihydrate equivalent to 500 mg amoxicillin and potassium clavulanate equivalent to 125 mg of clavulanic acid.
indicated for the treatment of the following infections in adults and children-
• acute bacterial sinusitis (adequately diagnosed)
• acute otitis media
• acute exacerbations of chronic bronchitis (adequately diagnosed)
• community acquired pneumonia
• cystitis
• pyelonephritis
• skin and soft tissue infections in particular cellulitis, animal bites, severe dental abscess with spreading cellulitis.
• bone and joint infections, in particular osteomyelitis.
consideration should be given to official guidance on the appropriate use of antibacterial agents.
posology
doses are expressed throughout in terms of amoxicillin/clavulanic acid content except when doses are stated in terms of an individual component.
the dose of augmentin that is selected to treat an individual infection should take into account:
• the expected pathogens and their likely susceptibility to antibacterial agents (see section 4.4)
• the severity and the site of the infection
• the age, weight and renal function of the patient as shown below.
the use of alternative presentations of augmentin (e.g. Those that provide higher doses of amoxicillin and/or different ratios of amoxicillin to clavulanic acid) should be considered as necessary (see sections 4.4 and 5.1).
for adults and children ≥ 40 kg, this formulation of augmentin provides a total daily dose of 1500 mg amoxicillin/375 mg clavulanic acid, when administered as recommended below. For children < 40 kg, this formulation of augmentin provides a maximum daily dose of 2400 mg amoxicillin/600 mg clavulanic acid, when administered as recommended below. If it is considered that a higher daily dose of amoxicillin is required, it is recommended that another preparation of augmentin is selected in order to avoid administration of unnecessarily high daily doses of clavulanic acid (see sections 4.4 and 5.1).
the duration of therapy should be determined by the response of the patient. Some infections (e.g. Osteomyelitis) require longer periods of treatment. Treatment should not be extended beyond 14 days without review (see section 4.4 regarding prolonged therapy).
shelf life
2 years in cold formed aluminium blisters (cfb) and desiccated pouch packs (dpp).
tablets in desiccated pouch packs should be used within 30 days of opening.